
Nitrogen
Nitrogen is one of the most demanded mineral elements by plants and as such one of the most critical inputs in farming. We have a range of mineral products, including urea, sulphate of ammonia and Nhance to aid nitrogen deficiencies and boost vegetative growth.
Urea
Features
Urea is an organic compound that occurs naturally and its synthetic version is the most widely used nitrogen fertiliser in the world. The wide acceptance of urea is due to its agronomic acceptability and low relative cost. Granular Urea is tailored for easy handling, even spreading and blending.
Uses
Plants take up nitrogen from the soil in the mineral forms of nitrogen, both ammonium and nitrate before converting it to plant protein nitrogen. Plants vary in their preference to utilise either form of nitrogen. Nitrate is considered the main source because it is mobile in the soil. On addition to the soil, urea dissolves into the soil solution and is converted to ammonium and then to nitrate.
Conversion is Favoured By:
- Temperature – the activity range is wide, varying from -20°C to 37°C with the optimum >20°C.
- Organic matter – activity is enhanced in soils high in organic matter which favours microbial activity.
- The time for urea to be converted to ammonium will depend on the conditions of moisture and temperature. The reaction will begin to occur 24 hours after application and be complete within 2 to 5 days. The reaction time is slowest in cold, waterlogged conditions.
Typical Analysis

S.O.A.
Features
Sulphate of ammonia is commonly used as a source of nitrogen and sulphur and is commonly blended with other fertilisers to supply sulphur. Sulphate features good storage and handling qualities and can also be readily blended with other fertilisers. Where conditions exist that nitrogen loses through volatilisation of Urea occur SOA can be used more efficiently as its nitrogen is in the ammonium form.
Uses
- SOA is a valuable fertiliser as a straight product or in blends.
- Sulphur in SOA is sulphate sulphur which is the form taken up directly by plants. Sulphate sulphur is readily soluble in water and goes straight into solution when applied to the soil (providing there is moisture).
- SOA releases nitrogen to the soil directly as ammonium NH4+, which is converted by bacteria to nitrate nitrogen. This process is favoured by conditions conducive to microbial activity, such as warm temperatures, moisture and organic matter. The rate of this reaction depends on those conditions and complete nitrification of applied ammonium can vary from a few days to several weeks.
Typical Analysis

Nhance
With fine particle fertiliser, the future of nitrogen application is here today.
Nhance is a fine particle fertiliser, providing the plant with tiny droplets of urea (or sulphate of ammonia when used), which can be assimilated across the plant leaf rapidly. By applying Nhance you’ll grow more grass, quicker.
Key Features & Benefits
- Environment – As Nhance is a fine particle fertiliser, less volume of nitrogen needs to be applied to achieve the equivalent results of granular products. The potential for farm related nitrate leaching is reduced whilst volatilisation of ammonia gas is almost eliminated.
- Animal Heath – By applying only 70% of the nitrogen compared to a typical urea application, the risk of nitrate poisoning is reduced and allows more flexibility within your paddock rotations.
- Return on Investment – Nhance directly impacts your bank account; by providing better returns than equivalent volumes of urea. The growth of dry matter in short periods of time ensures that the costs associated with supplementary feeds can be dramatically reduced.
- Soil – By applying less urea with every application, the soil acidification process is reduced. Urea itself is quite acidic and frequent applications over time will lead to a decrease in pH. By using fine particular fertiliser, you are slowing down the process firstly by using less urea and secondly by applying more to the leaf and avoiding soil contact.
- Time – With an application of Nhance, around 15% of the nitrogen lands on the leaf of the plant and the remaining nitrogen landing on the soil. By applying Nhance to the leaf, nitrogen uptake occurs within three hours from the application, which is enough nitrogen to feed the plant for ten days. By the time the leaf applied nitrogen has been used up, the remaining soil applied nitrogen has started working giving your grass consistent access to nitrogen.
With fine particle application, Gibsons Groundspread has the ability to apply solid and liquid fertilisers all-in-one pass, as well as Gibberellic Acid and chemicals so you can now grow grass and control weeds all in one application.
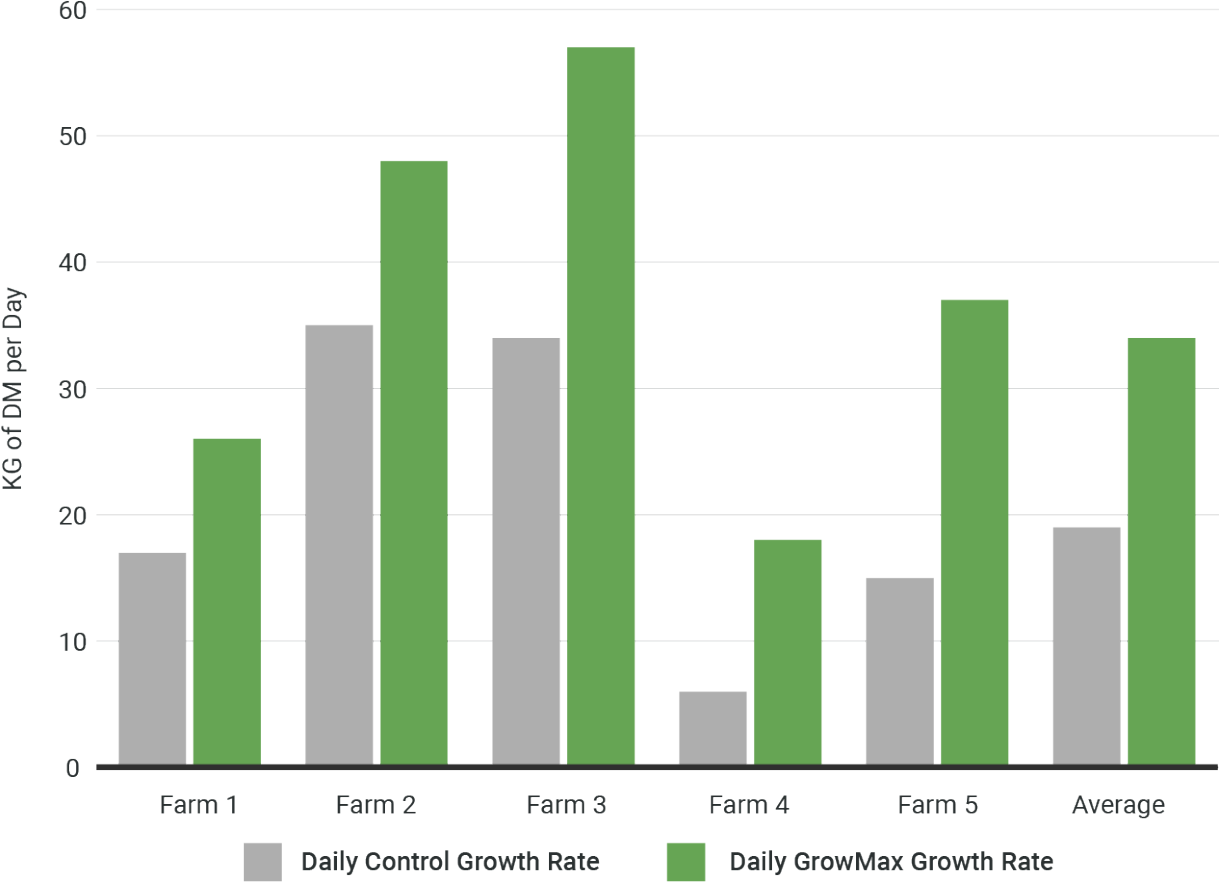
Trial Data Analysis
 Download brochure
Download brochure

 About Us
About Us
 History
History
 Team
Team
 Careers
Careers





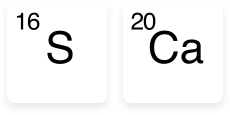

 Farm Mapping
Farm Mapping
 NPKS Reporting
NPKS Reporting
 Agronomy
Agronomy
 Soil Testing
Soil Testing
 Bin Delivery
Bin Delivery
 Spreading
Spreading
 Blending
Blending